Understanding the Reference Amounts Customarily Consumed (RACC) is crucial in the realm of food and beverage labeling, especially considering its significant impact on public health and consumer awareness. These guidelines, established by the Food and Drug Administration (FDA), serve as a standard for determining serving sizes on nutrition labels, ensuring consistency and clarity for consumers. The RACC is not just a regulatory requirement; it plays a pivotal role in guiding consumers to make informed dietary choices by providing a realistic representation of how much of a food item is typically consumed in one sitting.
This article will outline the intricacies of RACC, delving into its importance in creating accurate and FDA-compliant food labels. We will explore how RACC is determined, its application in various food categories, and its impact on both consumers and the food industry. By decoding the nuances of RACC, we aim to provide a comprehensive understanding of this key aspect of food labeling, highlighting its role in fostering informed consumer choices and promoting public health.
TLDR
- Reference Amounts Customarily Consumed (RACC) are FDA-established amounts typically eaten in one sitting.
- RACC is crucial for standardized serving sizes on nutrition labels.
- RACC and serving size are different; RACC is a regulatory standard while serving size is a practical, product-specific amount.
- FDA RACC guidelines categorize foods and beverages with specific reference amounts, including bakery products, beverages, cereals, dairy, fruits and vegetables, and snacks.
- Serving sizes are determined by identifying the product category, referring to the RACC value, considering package size, and adjusting based on consumption patterns.
- Single-serving containers contain amounts close to the RACC.
- Multi-serving containers exceed 200% of the RACC.
- FDA acknowledges products containing up to 300% of the RACC could be consumed in one sitting, requiring dual-column labeling.
- Exceptions to dual-column labeling include products requiring further preparation, commonly consumed with other foods, or specific categories like popcorn.
- FDA periodically updates RACC values to align with current eating habits.
- Changes in RACC affect serving sizes, %DV calculations, and nutrient content claims on labels.
- Regular review of FDA RACC guidelines is essential for label accuracy.
- Manufacturers should consult experts for products not fitting into RACC categories.
- Proactive label reviews are important for compliance, especially for new or reformulated products.
RACC Explained
What is RACC?
Reference Amounts Customarily Consumed (RACC) refers to the amount of a certain food or beverage that is typically eaten in one sitting. These amounts, established by the FDA, are based on data about the consumption habits of the general population. RACC is a critical component in the realm of food labeling, as it provides a standardized basis for determining serving sizes on nutrition labels. This standardization is essential for ensuring that consumers receive consistent and comparable nutrition information across different products. The FDA RACC guidelines encompass a wide range of food categories, each with a specific reference amount, reflecting the typical consumption patterns for each type of food or beverage.
The Difference Between RACC and Serving Size
While RACC and serving size are often used interchangeably, they are distinct concepts in food labeling. RACC, as we have stated, is a standardized amount that serves as a reference for labeling purposes. It represents an average portion that people typically consume, not necessarily a recommended amount for consumption. In contrast, the serving size on a food label is the specific amount of food that the manufacturer suggests as a single serving. This serving size is often based on the RACC but can be adjusted by the manufacturer to reflect the packaging and marketing strategy of the product.
For instance, if the FDA RACC for a snack food is 30 grams, a manufacturer might choose a serving size of 15 grams for a smaller package or 60 grams for a larger one. The key difference lies in the purpose: RACC is a regulatory standard for consistency in labeling while serving size is a practical representation tailored to each specific product. Understanding this distinction is crucial for consumers to accurately interpret nutrition labels and make informed dietary choices.
See How FoodLabelMaker Can Help You
Product Categories and Their Reference Amounts
The FDA RACC guidelines categorize a wide array of food and beverage products, each with its own set reference amount. These categories and their associated RACC values play a pivotal role in ensuring consistency and clarity in food labeling. Here’s a look at some common product categories and their corresponding RACC values:
- Bakery Products: For example, bread typically has a RACC of 50 grams.
- Beverages: The RACC for most beverages, like soft drinks, is 360 milliliters.
- Cereals and Other Grain Products: A standard RACC for dry, instant cereals is around 15 grams.
- Dairy Products: For instance, the RACC for yogurt is set at 170 grams.
- Fruits and Vegetables: A common RACC for dried fruit like dried apples is 40 grams.
- Snacks: Snack foods like chips often have a RACC of 30 grams.
These categories and their RACC values are integral in guiding manufacturers and consumers alike in understanding portion sizes and nutritional content.
Using RACC to Determine Serving Sizes
Translating FDA RACC values into serving sizes for food products requires careful consideration of several factors. Here’s a step-by-step guide to help manufacturers and consumers alike in determining appropriate serving sizes:
- Identify the Product Category: Determine which FDA RACC category your product falls into. This is the starting point for establishing a serving size.
- Refer to the RACC Value: Consult the FDA’s RACC chart to find the reference amount for your product category.
- Consider the Package Size: If your product’s package size is close to the RACC, it’s often practical to use the RACC as the serving size. However, if the package size significantly differs from the RACC, adjustments may be necessary.
- Adjust Based on Consumption Patterns: Consider how people typically consume the product. If it’s commonly eaten in smaller or larger quantities than the RACC, adjust the serving size accordingly.
- Ensure Clarity and Honesty: The serving size should reflect a realistic quantity that consumers are likely to eat, ensuring that the nutritional information is both useful and honest.
By following these steps and considering factors like package size and consumption habits, manufacturers can determine serving sizes that are both compliant with FDA regulations and helpful for consumers in making informed dietary choices.
Single-serving vs. Multi-serving Containers
Understanding the distinction between single-serving and multi-serving packages is crucial in food labeling and consumer awareness. The FDA RACC guidelines also play a significant role in this classification. Here’s how:
- Single-serving Containers: These are packages that contain an amount very close to the RACC. If a product’s total amount is less than or equal to 200% of the RACC, it is considered a single-serving container. This means the entire contents of the package can reasonably be consumed in one sitting. There is an exception for this as the FDA acknowledges that certain products, containing up to and including 300% of the RACC, could also feasibly be consumed by an individual on a single eating occasion and would therefore be required to bear dual-column labeling.
- Multi-serving Containers: Conversely, if a product’s total amount exceeds 200% of the RACC, it is classified as a multi-serving container. These packages contain multiple servings, and the label must clearly indicate the number of servings per container, with each serving size being based on the RACC for that category.
The FDA RACC values are instrumental in this classification, ensuring that consumers have a clear understanding of whether a package is meant to be consumed in one sitting or if it contains multiple servings.
Dual-Column Labeling
The dual-column labeling approach, directly influenced by FDA RACC values, is designed to provide consumers with more comprehensive nutritional information. This labeling format is essential for products containing between 200% to 300% of the RACC, which could be consumed in a single eating occasion.
In this approach, food package labels display two columns of nutritional information. The first column lists the nutritional facts for a single serving, as determined by the RACC, while the second column shows information for the entire package. Exceptions to this requirement include products that require further preparation, are commonly consumed with other foods, or fall into specific categories like popcorn, where different labeling formats are permissible.
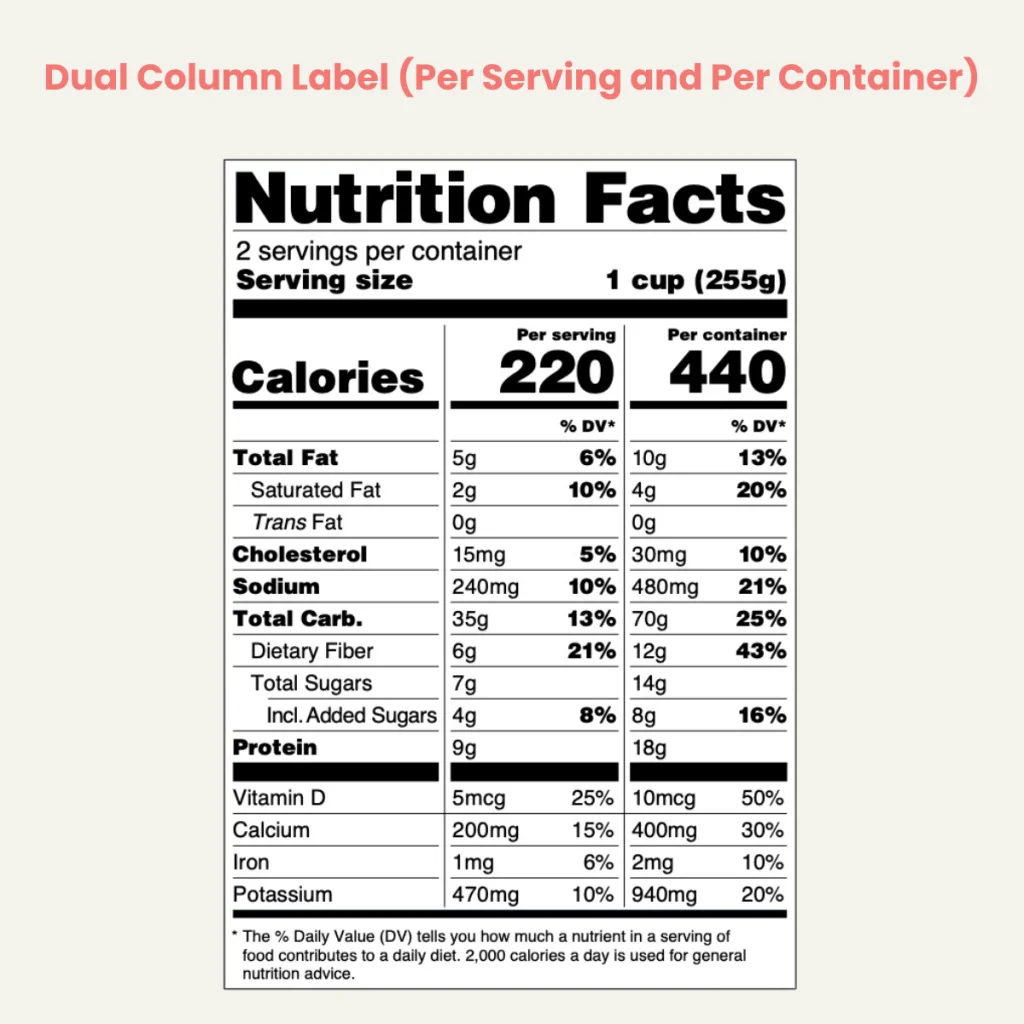
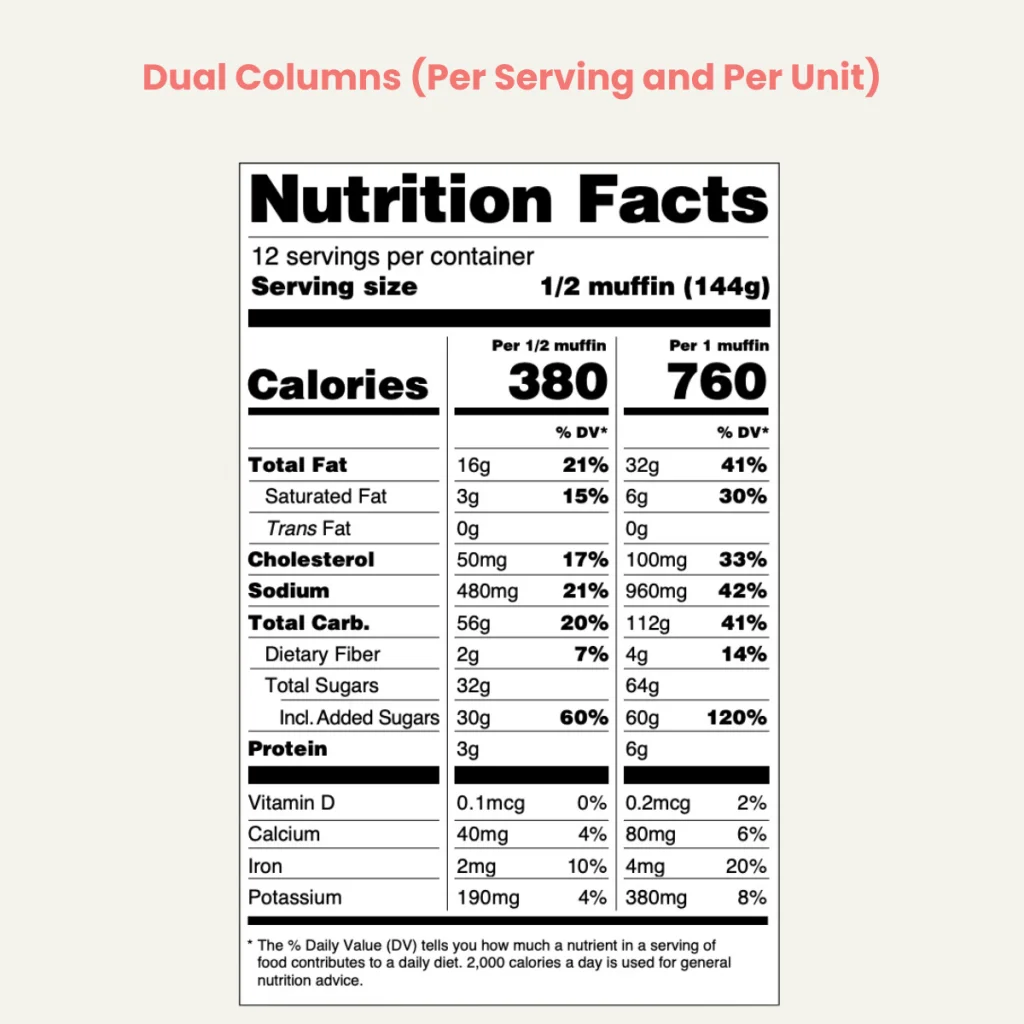
For discrete units within the 200 to 300 percent RACC range, the label must provide dual-column information, unless an exemption applies. This dual-column format helps consumers understand the nutritional content of both a single serving and the entire package, aiding in more informed dietary choices.
Recent Changes in RACC and Their Impacts
The FDA periodically reviews and updates RACC values to reflect changes in eating habits and new nutritional science. These updates can have significant implications for food labeling:
- Updates in RACC Values: Recent changes in FDA RACC values have been made to better align with how people eat today. For instance, the RACC for ice cream was previously set at 1/2 cup but has been updated to 2/3 cup, reflecting more realistic consumption patterns.
- Implications for Serving Sizes: These updates directly impact the serving sizes listed on food labels. A change in the RACC value may lead to a corresponding change in the declared serving size, ensuring that it remains a realistic representation of how much people typically eat or drink.
- Effects on %DV Calculations and Nutrient Content Claims: Changes in RACC values also affect the Percent Daily Values (%DV) calculations and nutrient content claims. As serving sizes change, the %DV for nutrients in a serving will also change, potentially altering how a product meets certain nutrient content claims like “low fat” or “high fiber.”
These updates in FDA RACC values are crucial for ensuring that food labels provide consumers with the most relevant and current information regarding serving sizes and nutritional content.
Best Practices and Recommendations
For food manufacturers and brands, adhering to FDA RACC guidelines is a crucial aspect of ensuring label accuracy and regulatory compliance. One key practice is the regular review of these guidelines. This ongoing process ensures that product labels accurately reflect the most current serving size requirements, including any updates to RACC values. Adjusting product labels in line with these changes is an essential step in maintaining compliance.
Another challenge that manufacturers frequently encounter is determining the appropriate serving size for products that don’t clearly fit into an established RACC category. In such situations, it’s beneficial to seek guidance from food labeling experts or consult directly with the FDA. This approach helps in identifying the most fitting serving size based on the closest relevant RACC category.
Additionally, conducting proactive reviews of product labels is vital, especially when introducing new products to the market or reformulating existing ones. Such reviews are important because changes in ingredients or package size can impact the applicable RACC value, necessitating adjustments to serving sizes on labels.
By embracing these best practices and keeping abreast of FDA RACC guidelines, manufacturers can effectively manage the complexities of food labeling. This diligence ensures that their products not only remain compliant but also provide clear and transparent information to consumers.



