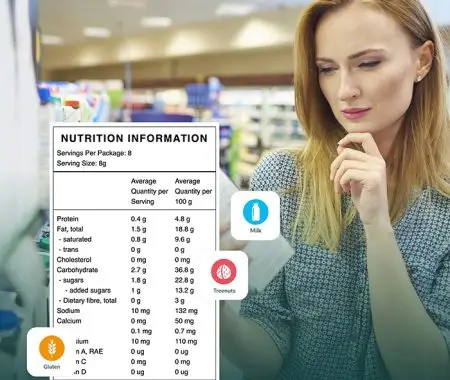
The ingredients list is one of the first things a food safety auditor, retailer buyer, or customs officer looks at when picking up a product. Getting it wrong can lead to potential recalls, delisting, and in serious cases, enforcement action from FSANZ or a state food authority.
Under the Food Standards Australia New Zealand Code (FSANZ), the rules for declaring ingredients are more detailed than most food businesses realize. Standard 1.2.4 details how an ingredients list must be structured, including what order ingredients appear in, how they are named, and when compound ingredients need to be broken down into their individual components. Standard 1.2.10 adds another layer: if a particular ingredient is emphasised through the product name, imagery, or marketing, the manufacturer is required to declare what percentage of that ingredient is actually in the product.
This article covers both standards in full, with worked examples to show exactly what compliant labelling looks like in practice.
Already know your allergen obligations? If not, read the guide on Allergen Labelling & Mandatory Declarations first. Allergen declarations sit within the ingredients list, so understanding both together is essential.Get started with Food Label Maker to create FSANZ-compliant nutrition labels.
See How FoodLabelMaker Can Help You
FSANZ Statement of Ingredients Explained (Standard 1.2.4)
Standard 1.2.4 sets out the rules for how ingredients must be listed on a food label in Australia and New Zealand, including the order they appear in, how they are named, and when sub-ingredients must be declared.
When Is A Statement Of Ingredients Required?
A statement of ingredients is generally required for any food for sale and must list each ingredient present in the food.
However, there are a few exceptions where a statement of ingredients is not required:
- If the label already lists the name of the food, and a statement of ingredients would only consist of that exact name, the label itself is considered to contain the required statement.
- For example, a package of plain rolled oats or a bag of white sugar does not need a formal ingredients list. The product name alone communicates the full composition to the consumer.
- Water that is packaged and labelled in accordance with Standard 2.6.2.
- Standardised alcoholic beverages.
- Food for sale that is contained in a small package.
- According to Standard 1.1.2, small packages are defined as: a package with a surface area of less than 100 cm2.
How Ingredients Must Be Ordered on an FSANZ Food Label
Under Standard 1.2.4, ingredients must be listed in descending order of ingoing weight – from greatest to least at the time of manufacture, not based on the finished product. This distinction matters because ingredients change during cooking and processing. Water added during production may largely evaporate during baking due to moisture loss, but its ingoing weight still determines its position in the list.
It’s important to note that if a dehydrated or concentrated ingredient is used, the rules differ depending on the purpose: for ordering the ingredients list, the weight before dehydration or concentration may be used; for calculating characterising ingredient percentages under Standard 1.2.10, the rehydrated weight must be used instead.
How to Name Ingredients and Food Additives on an FSANZ Ingredients List
Under Standard 1.2.4, each ingredient must be declared using its common or usual name, meaning the name that consumers would recognize and expect to see. Generic descriptions or vague terms are not acceptable where a recognized name exists. For example, “dairy product” is not acceptable where “cheddar cheese” or “skim milk powder” would be the recognised name.
In some cases, a class name may be used in place of a specific ingredient name. Permitted class names include “vegetable oil”, “spices”, “herbs”, “starch”, “flour”, “sugar”, and “fish”, among others. So a product containing sunflower oil may list it as “vegetable oil”, and a product containing oregano, thyme, and rosemary may list them collectively as “herbs”. Class names are only permitted where the standard explicitly allows them, so a manufacturer cannot simply group ingredients under a class name for convenience.
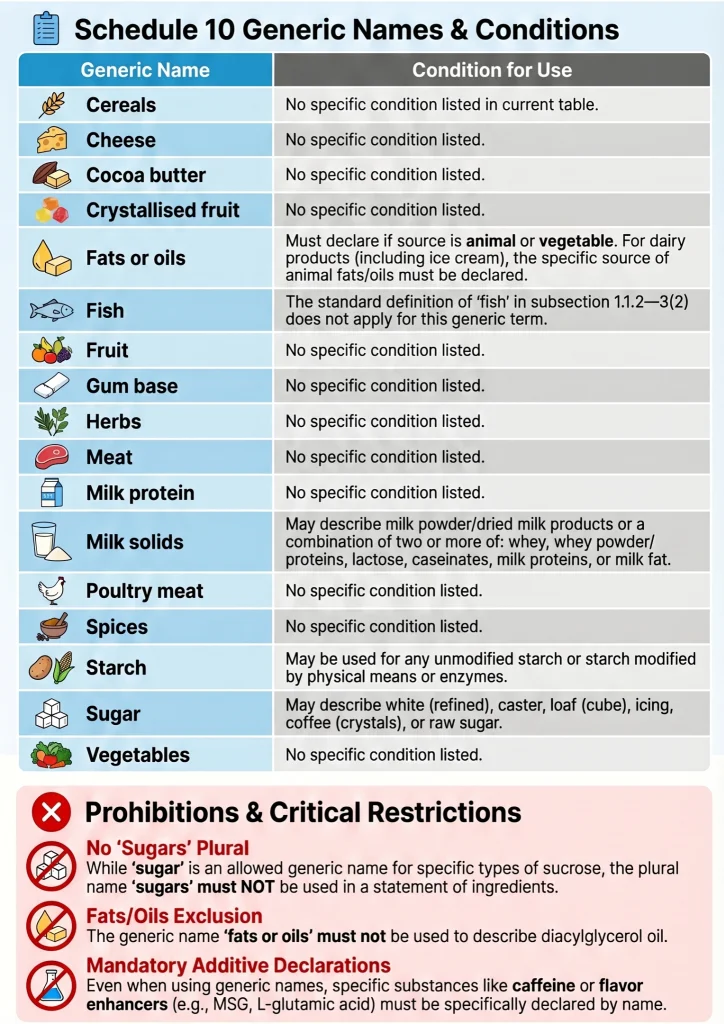
Food additives follow a different naming convention. They must be declared using the additive class name followed by either the specific additive name or its code number in brackets. For example, a preservative may be declared as “Preservative (220)” or “Preservative (Sulphur Dioxide)”. A mineral salt may be declared as “Mineral Salt (451)” or “Mineral Salt (Potassium Tripolyphosphate)”. The additive class name must always be present — listing a code number alone without the class name is not compliant.
Flavours are a common exception. Natural and artificial flavours may be declared simply as “Natural Flavour” or “Artificial Flavour” without specifying the source, unless the flavour is derived from a common allergen — for example, a natural flavour derived from milk must declare that source.
Compound Ingredients: When To List Sub-Ingredients on FSANZ Labels
A compound ingredient is any ingredient that is itself made up of two or more ingredients. Soy sauce, for example, is a compound ingredient because it contains water, soybeans, wheat, and salt. Tomato paste, stock, and bread are all compound ingredients for the same reason.
Under Standard 1.2.4, when a food contains a compound ingredient, the sub-ingredients that make up that compound ingredient must generally be declared in the statement of ingredients. This is because consumers have the right to know what is actually in the food, not just the name of a processed ingredient that contains several components.
However, there is an important threshold. If a compound ingredient makes up less than 5% of the finished food, the sub-ingredients of that compound ingredient do not need to be individually declared. The compound ingredient can simply be listed by its own name. This is commonly referred to as the 5% rule.
There is one exception that overrides the 5% rule entirely. If any sub-ingredient of a compound ingredient is a declared allergen under Standard 1.2.3, it must be declared regardless of what percentage the compound ingredient represents in the finished product. The 5% threshold provides no exemption from allergen declaration obligations.
A practical example: a stir-fry sauce contains soy sauce at 3% of the finished product. Because soy sauce is below the 5% threshold, its sub-ingredients (water, soybeans, wheat, and salt) do not need to be individually listed. However, soybeans and wheat are both declared allergens. Both must still be declared in the ingredients list despite the compound ingredient falling below 5%.
How to Handle Undisclosed Sub-Ingredients From Suppliers
When a food business purchases an ingredient from a supplier, that ingredient may itself contain sub-ingredients that are not immediately obvious from the product name alone. A flavouring paste, a spice blend, or a premixed sauce base may contain additives, allergens, or components that the manufacturer was not aware of at the time of formulating the product. Under Standard 1.2.4, those sub-ingredients must still be declared on the finished product label if they are required to be listed.
This places an active due diligence obligation on food manufacturers. It is not sufficient to rely on the common name of a supplier ingredient and assume its composition is simple. Manufacturers are expected to obtain full ingredient declarations from their suppliers and review them to determine whether any sub-ingredients trigger a declaration requirement on the finished label, particularly in relation to allergens.
In practice, this means requesting a full specification sheet or ingredient breakdown from every supplier, reviewing those documents each time a supplier changes their formulation, and updating the finished product label accordingly.
A practical example: a manufacturer sources a pre-made curry paste and lists it simply as “curry paste” on their label. The supplier later changes their formulation to include a peanut-based thickener. If the manufacturer has not requested updated specifications, they may unknowingly be selling a product containing peanuts without an allergen declaration, which is a serious compliance failure and potential safety risk. The responsibility for declaring that allergen sits with the finished product manufacturer, not the supplier.

When Does Water Need to Be Declared on an FSANZ Ingredients List?
Water added during the manufacture of a food must generally be declared in the statement of ingredients. It is treated as an ingredient in its own right and listed in its correct position according to ingoing weight, in the same way as any other ingredient.
There are several situations in which water does not need to be declared in the statement of ingredients:
- It constitutes less than 5% of the finished food
- It forms part of a broth, brine, or syrup that is already declared in the statement of ingredients or forms part of the name of the food
- It is added specifically to reconstitute dehydrated or concentrated ingredients
- It is driven off during cooking, baking, or processing and is not meaningfully present in the finished product
A practical example: a bread manufacturer adds water as a primary ingredient during mixing. That water remains largely present in the finished loaf and must be declared. By contrast, a manufacturer who adds water solely to reconstitute a dehydrated vegetable powder during production is not required to declare that water separately.
It is worth noting that water packaged and labelled in accordance with Standard 2.6.2 (regulations for non-alcoholic beverages and brewed soft drinks) is exempt from requiring an ingredients statement altogether. This is a labelling exemption that applies to the product itself rather than a rule about declaring water as an ingredient.
What Are The FSANZ Ingredients List Formatting Requirements?
Standard 1.2.4 does not prescribe specific formatting or placement rules for the statement of ingredients. There is no mandated font size, no required panel position, and no specified layout for how the ingredients list must visually appear on a label.
What the standard does govern strictly is the content of the ingredients list — the names used, the order ingredients appear in, and when percentages must be declared. How that content is visually presented falls under the broader labelling requirements in Standard 1.2.1 and the general legibility provisions that apply to food labels overall. These require that mandatory label information be presented in a way that is legible and comprehensible to consumers under normal conditions of purchase.
In practice this means the ingredients list must be readable and cannot be printed in a font so small or in a colour so low in contrast that a consumer could not reasonably read it.
FSANZ Characterising Ingredients Explained (Standard 1.2.10)
Standard 1.2.10 requires food businesses to declare the percentage of any ingredient that is emphasised through the product name, imagery, or marketing, ensuring consumers know exactly how much of a highlighted ingredient is actually in the product.
What Is an FSANZ Characterising Ingredient and When Must a Percentage Be Declared?
A characterising ingredient is any ingredient or component of a food that is emphasised or implied on the food label in a way that is likely to influence a consumer’s purchasing decision.
According to the Australia New Zealand Food Standards Code 1.2.10, for an ingredient to be considered a characterising ingredient, it must meet the following criteria:
- It is mentioned in the name of the food, for example “Strawberry Yoghurt” or “Chicken and Vegetable Pie”
- It is usually associated with the name of the food by a consumer, for example “Butter Chicken Sauce”
- It is emphasised on the label of the food through words, pictures, or graphics, for example a product that features a prominent image of salmon on the front panel.
Where an ingredient meets any of these criteria, the food business must declare the percentage of that ingredient in the finished product. This is commonly referred to as QUID — Quantitative Ingredient Declaration. The underlying principle is straightforward: if an ingredient is being called out or implied, consumers have the right to know exactly how much of it is present in the product.
QUID Calculation: How to Calculate Your Characterising Ingredient Percentage
The proportion of a characterising ingredient (PCI) is calculated using the following formula:
PCI = IW ÷ TW
Where IW is the ingoing weight of the characterising ingredient and TW is the total weight of all ingoing ingredients.
Four rules modify this base calculation:
Added water and volatile ingredients — the weight of added water or volatile ingredients removed during manufacture must not be included in TW when calculating PCI. This prevents the percentage from being artificially diluted by water no longer present in the finished product.
Reconstituted ingredients — if a concentrated or dehydrated ingredient is reconstituted during manufacture, the reconstituted weight of that ingredient may be used as IW rather than its dry weight.
Foods requiring reconstitution before consumption — if the finished product requires reconstitution before consumption, such as a powdered drink mix, PCI may be calculated as a proportion of the food as reconstituted rather than as sold.
Moisture loss during processing — where moisture loss occurs during processing, the manufacturer may alternatively calculate the characterising ingredient percentage based on the actual weight of that ingredient in the finished product rather than its ingoing weight. This is a permitted alternative, not a mandatory method.
As an example, here’s a step-by-step worked example using a Blueberry Muffin:
Assume the following ingoing weights:
- Flour: 500g
- Sugar: 200g
- Butter: 150g
- Eggs: 100g
- Blueberries: 120g
- Milk: 80g
- Baking powder: 10g
Total ingoing weight: 1,160g. Water and volatile ingredients lost during baking must be excluded from TW. Assuming 80g of moisture is lost during baking, TW = 1,080g.
Blueberry percentage: 120 ÷ 1,080 × 100 = 11.1%, rounded to 11%.
The label must therefore declare blueberries at 11%, placed immediately after the ingredient name in the ingredients list.
Displaying QUID Percentages: Placement and Rounding Rules
The percentage must be declared as a whole number rounded to the nearest whole number. Where the percentage is below 5%, it may be rounded to the nearest 0.5 decimal place — for example 3.5% rather than 4%.
The percentage may be placed in one of two locations: immediately following the ingredient name in the statement of ingredients, or in or adjacent to the product name on the label. Both are valid under the standard. In practice, placing the percentage within the ingredients list is the more common approach as it keeps all ingredient information in one place and is easier to maintain when recipes change.
FSANZ Ingredients List Compliance: Key Takeaways
Getting the ingredients list right is one of the most detail-intensive parts of food labelling compliance. Standard 1.2.4 governs the content and structure of the statement of ingredients — from the order ingredients appear in, to how they are named, to when sub-ingredients of compound ingredients must be declared. Standard 1.2.10 adds a further layer of obligation for any ingredient that is emphasised through the product name, imagery, or consumer expectation, requiring a percentage declaration that accurately reflects how much of that ingredient is present in the finished product.
The most common area of non-compliance is not understanding where the rules intersect. Allergen declarations sit within the ingredients list and cannot be omitted regardless of compound ingredient thresholds. Characterising ingredient percentages must account for moisture loss and reconstitution. Supplier formulation changes can create undeclared allergens overnight if due diligence processes are not in place.
For food businesses, the safest approach is to treat the ingredients list not as a formatting exercise but as a live compliance document that must be reviewed every time a recipe, supplier, or process changes.
Frequently Asked Questions About FSANZ Ingredients Lists
Do I need to list water if it evaporates during cooking?
No. UnderStandard 1.2.4, water that is driven off during cooking, baking, or processing and is not meaningfully present in the finished product does not need to be declared in the statement of ingredients. However, if water remains present in the finished product in a meaningful quantity, it must be declared in its correct position according to ingoing weight.
Ready to create a complaint FSANZ nutrition information panel? Get started now.
Does a picture of a fruit on my packaging trigger a QUID declaration?
Yes, it can. UnderStandard 1.2.10, an ingredient that is emphasised through pictures or graphics on the label is a characterising ingredient and requires a percentage declaration. A prominent image of strawberries on a product that contains a token amount of strawberry flavouring would trigger a declaration obligation regardless of whether the word “strawberry” appears in the product name.
Get started here and create nutrition labels ready for the Australia and New Zealand food market.
My characterising ingredient is less than 1% of the finished product — do I still need to declare it?
Yes. There is no minimum threshold below which a characterising ingredient percentage declaration is not required. If an ingredient meets the criteria for a characterising ingredient underStandard 1.2.10, its percentage must be declared regardless of how small that percentage is. Where the percentage is below 5%, it may be rounded to the nearest 0.5 decimal place rather than the nearest whole number.
Explore our nutrition label software and get started on your FSANZ label today.
Can I use a class name for an ingredient that contains an allergen?
No. WhileSchedule 10 permits the use of generic class names for certain ingredient categories, allergen declaration obligations underStandard 1.2.3 always override class name permissions. If an ingredient within a permitted class name contains or is derived from a declared allergen, that allergen source must be specifically declared regardless of whether the class name would otherwise be permitted.
Not sure how to get started? Reach out to one of our regulatory experts.
What happens if my supplier changes their formulation?
The responsibility for accurate allergen and ingredient declarations on the finished product sits with the manufacturer, not the supplier. If a supplier changes their formulation and introduces a new sub-ingredient — particularly an allergen — without notifying the manufacturer, the finished product label may become non-compliant under Standard 1.2.4. This is why requesting updated specification sheets from suppliers regularly and reviewing them against current labels is an essential part of ongoing compliance. A reactive approach to supplier changes is one of the most common causes of allergen-related recalls in Australia.
Explore our pricing plans here or get started with your FSANZ label today.